Food producers and importers are allowed to promote the nutritional and health benefits of their foods, provided that they comply with the EU rules on labelling, presentation and advertising.
In practice, however, this often goes wrong, for instance in the form of misleading claims, making products appear healthier than they actually are. A great deal of public attention is being paid to this (see, for instance, here, here and here). The Jumbo supermarket chain, for instance, was recently ‘awarded’ the Gouden Windei (Golden Wind Egg) for the most misleading food product of 2022: its three ‘healthy’ Ice Teas.
Various obligations apply to the use of nutrition and health claims on foods. The labels of food products must state the name of the food, the ingredients and the use-by date. And if a company uses nutrition and health claims, they may in no event be misleading, confusing or ambiguous. Additional rules furthermore apply to the voluntary indication of nutrition and health claims. Failure to comply with these laws and regulations can lead to high fines being imposed by the Netherlands Food and Consumer Product Safety Authority (“NVWA”).
Claims: voluntarily provided food information
First, a nutrition or health claim must be correctly qualified. Second, it must be established whether the claim is legally permitted, and various laws and regulations must be met. In some cases, a European authorisation procedure must first be completed.
Regulation 1169/2011 sets out various rules on the provision of food information to consumers and on food labelling, the basic principle being that food information may not be misleading, confusing or ambiguous.
Claims, such as health claims and nutrition claims, are a form of voluntarily provided food information. A claim provides information about a food product. This applies to any message about certain properties of a food product. In addition to the text on the packaging, this includes pictures, illustrations and symbols that imply something about the product’s composition or its health effect, for instance commendations such as ‘healthy’ or stating that a product is ‘packed with vitamins’.
The use of health and nutrition claims is further regulated at a European level in Regulation 1924/2006 on nutrition and health claims made on foods (“the Claims Regulation”). The Claims Regulation applies to nutrition and health claims used in commercial communications about food products intended to be sold to consumers as such.
Nutrition claim
A nutrition claim is a claim that states, suggests or implies that a food product has certain positive active nutritional properties relating to energy value (calorific value) or nutrients, or other substances that it does or does not contain. Examples include claims about the properties of the product, such as ‘low-fat’, ‘sugar-free’, ‘high-fibre’ and ‘low in saturated fats’. Food manufacturers may use a nutrition claim only if it is mentioned in the list of authorised claims mentioned in the Annex of the Claims Regulation. The food product must meet certain requirements in order to carry a claim. The ‘light’ claim, for instance, may be used only if the product contains 30% less sugars than a comparable product.
Health claim
A health claim is a claim that states, suggests or implies a (positive) relationship between a food category, food product or component and the consumer’s health. Health claims often relate to physiological effects but may also pertain to psychological and slimming effects, or to disease risk reduction. A case in point is a claim such as ‘potassium contributes to normal function of the muscles’ or ‘magnesium helps reduce fatigue’.
Health claims are permitted, provided that they comply with general and specific regulations for health claims, have been authorised after a European authorisation procedure, and have subsequently been included as an authorised claim in the European Claims Register. The Claims Database applies specifically to the Netherlands.
No properties may be attributed to food in the form of a claim regarding the prevention, treatment or cure of a human disease. If a food product is the subject of a medical claim, that product falls under the definition of a medicine (and therefore under the Geneesmiddelenwet (Medicines Act)). The ban on medical claims relating to a food does not apply to disease risk reduction claims used in accordance with the conditions set out in the Claims Regulation.
Authorisation of health claim
A food manufacturer may use a health claim only if it has been authorised (Regulation 353/2008 on the authorisation of health claims and Regulation 907/2013 on the use of generic descriptors). For each individual health claim, a separate application must be submitted to the competent authority of a Member State. In the Netherlands, that authority is the NVWA. The NVWA then forwards that application to the European Food Safety Authority (“EFSA”), based in Italy. The EFSA advises on the scientific substantiation underlying a health claim. It critically reviews the substantiation of the claims and then forwards its advice to the European Commission (“the Commission”). The Commission then approves or rejects the claim and publishes its decision in a regulation in the Official Journal of the European Union. The application process takes a total of about eight months.
Authorised claims are entered in the European Claims Register, which contains an overview of which nutrition and health claims are permitted under which conditions. Subject to the applicable conditions, authorised claims may be used by any food business operator (also if it was applied for by another party). The register also states which claims have been rejected. In addition to the claim authorisation procedure, food companies may also submit their nutrition or health claims to a preliminary reviewed by the Keuringsraad Kennis en Advies Gezondheidsreclame (Dutch Inspection Board for Knowledge of and Advice on Health Advertising).
There are also what is known as ‘on hold’ health claims. These are health claims whose assessment has not yet been completed at a European level. The use of an ‘on hold’ health claim requires a substantiation based on generally accepted scientific evidence.
Enforcement of nutrition and health claims
The NVWA is in charge of supervising food safety and may take enforcement action in the event of violations, for instance by imposing fines and orders subject to penalties. In 2018, for instance, it investigated 36 parties in the food industry engaged in weight-loss diets. In that investigation, the NVWA established at 21 of the 36 companies violations relating to nutrition claims and at 28 of the 36 companies violations relating to health claims. Later that year, the NVWA investigated a further 132 different breakfast cereals that stated at least one nutrition or health claim. Of these, 22 products were misleading, as a result of which the packaging had to be changed.
In 2022, the NVWA imposed a fine of over €73,000 on an online drugstore. The drugstore sold products making claims about health benefits for muscles and tendons that are not permitted by law. The online drugstore continued to state the claims on the products, despite repeated warnings and fines from the NVWA.
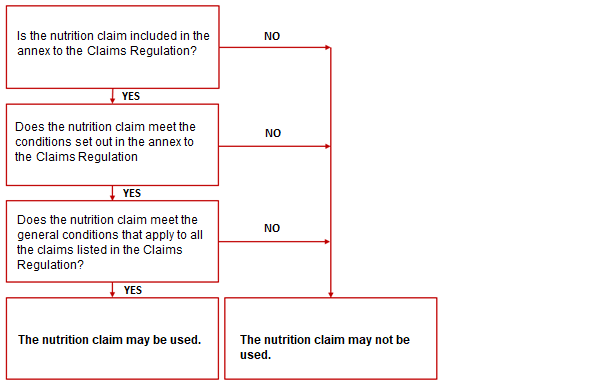
Checklist when making a nutrition claim
Checklist when making a health claim unrelated to disease risk reduction
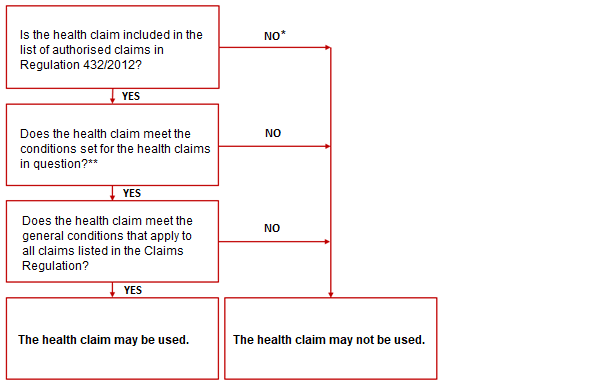
*The health claim may still be ‘on hold’ in the European Claims Register.
**See Regulation 432/2012 or the European Claims Register.
Checklist when making a health claim related to disease risk reduction or children
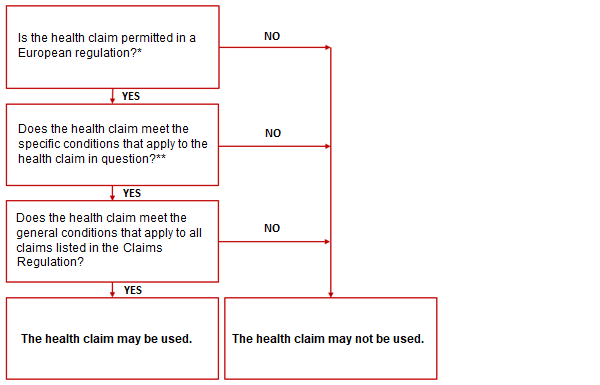
*See the European Claims Register.
*See the conditions set out in the regulation under which the claim is authorised.
Please do not hesitate to contact us if you have any questions about nutrition and health claims.
Follow Maverick Advocaten on LinkedIn







